|
The Pauli Exclusion Principle in quantum mechanics forbids electrons (and all fermions with half integer spin including neutron) occupying the same state. Basically, each electron must have different energy when they are packed together, as they are in a white dwarf. The number of available low energy states is too small and many electrons are forced into high energy states. When this happens the electrons are said to be degenerate. These high energy electrons make a significant contribution to the pressure. Because this pressure arises from a quantum mechanical effect, it is insensitive to temperature, i.e., the pressure doesn't go down as the star cools. This pressure is known as electron degeneracy pressure and it is the force that supports white dwarf stars against their own gravity. If the star is more massive, the Fermi energy (corresponding to the last-filled energy state at T = 0 K) goes up and it becomes possible | |
to absorb the electrons into the nucleons, converting protons into neutrons. In this case the Fermi energy reaches to a level above 1 MeV. If the electrons disappear this way, the star collapses suddenly down to a size for which the degeneracy pressure of the neutrons stops the collapse (with quite a shock). Some white dwarfs stay at earth size for a long time as they suck in mass from their surroundings. When they have acquired enough mass, they collapse to form a neutron star and explode as supernova. These are the Type Ia supernovae, which produce nearly same amount of energy. Thus, they have been used to measure the distance of astronomical objects, leading to the discovery of accelerating expansion of the universe. It is possible that there are other kinds of degeneracy stars as the system is stablized by successive layers of constituent particles (see Table 01 below).
|

 12
12  0, if state a is the same as state b. In other words, the two fermions cannot occupy the same energy level if their spin state and position are also equal. The two particles can be considered as practically coincided if their separation is less than the unresolvable limit imposed by the uncertainty principle. The Fermi-Dirac distribution for a system of fermion gas (Figure 01) shows that for a certain position and spin state (either up or down), at most one fermion can occupy certain energy. They fill up all those energy levels up to the Fermi energy EF for T = 0, and spill
0, if state a is the same as state b. In other words, the two fermions cannot occupy the same energy level if their spin state and position are also equal. The two particles can be considered as practically coincided if their separation is less than the unresolvable limit imposed by the uncertainty principle. The Fermi-Dirac distribution for a system of fermion gas (Figure 01) shows that for a certain position and spin state (either up or down), at most one fermion can occupy certain energy. They fill up all those energy levels up to the Fermi energy EF for T = 0, and spill
 0. It is these high energy fermions that generate the degeneracy pressure as the fermions are squeezed into a small volume.
0. It is these high energy fermions that generate the degeneracy pressure as the fermions are squeezed into a small volume.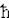 2/2m) (3
2/2m) (3 2N/V)2/3, where V is the volume, N the total number of fermions, and m the mass of the fermion. Thus from the known value of the number density n = N/V, we can estimate EF, the inter-fermion separation
2N/V)2/3, where V is the volume, N the total number of fermions, and m the mass of the fermion. Thus from the known value of the number density n = N/V, we can estimate EF, the inter-fermion separation  d
d  (2
(2