One characteristic shared by all species is the accumulation of unrepaired somatic damage. Thus, lifelong accumulation of various types of damage; along with random errors in bio-informational processes are the major causes of ageing. Interventions that
 |
 |
remove damage might counter some of the adverse effects. But macromolecular damage comes in many forms (Figure 05b), many of which have yet to be identified. It might seem more efficient to eliminate damaging molecules, rather than the damage itself. However, some damaging molecules are crucial for normal cellular function we cannot do without. Some time interventions are two edges sword. For example, tissue regeneration elevates cancer risk by increasing the chance of acquiring DNA mutations or epi-mutations. |
Figure 05a Causes of Ageing [large image] |
Figure 05b Macromolecular Causes [view large image] |
While tumour suppressor mechanisms either eliminate cells that acquired extensive damage (apoptosis) or permanently prevent their proliferation (senescence). |
 |
|
Figure 05c DNA Repair |
Aging is probably the result of the breakdown in the cellular safety nets - not enough anti-oxidants are produced naturally. In addition, modern life has enormously increased the number of toxic free-radicals introduced into our bodies every day from the
 |
 |
 |
surrounding environment. The most significant sources of excess free-radicals are dietary or environmental. Dietary sources are usually fats, either rancid or hydrogenated fats, and fats that have been heated to high temperatures during cooking. Environmental pollutants include automobile exhaust, cigarette smoke and numerous other chemicals, physical exercise, stress, and radiation. Table 04 summarizes the variety of defenses to prevent or repair molecular damage caused by free radicals, but as a group they are thought to be imperfect. Some evidence indicates that certain of the defenses also become less effective over time (ageing). New study found that in the course of ageing, certain people are more vulnerable than others to age-related damage from ROS, and genes related to memory and learning appear to be more vulnerable than other genes. Further research is needed to identify the causes. |
Figure 06a ROS |
Figure 06b SOD |
Table 04 Defenses |
 |
Lately in 2008, it is suggested that drinking or eating isotopes can slow down the chemical reactions with ROS. It is because the heavy isotopes form stronger covalent bonds than their lighter counterparts. While the effect applies to all heavy isotopes, including those with the elements of life such as carbon-13, nitrogen-15 and oxygen-18, it is most marked with deuterium (heavy water with one extra neutron) as it is proportionally so much heavier than hydrogen. Deuterated bonds can be up to 80 times stronger than those containing hydrogen (Figure 06). The idea has been tested on fruit flies. It is found that large dosages were deadly, smaller quantities increased lifespans by up to 30%, and herein lies some problems with this big idea: |
Figure 06c ROS Damage Control [view large image] |
- Safety - Although carbon-13 and the other elements of life seem to be essentially non-toxic, the tolerant limit for deuterium in mammals is at about the 20% mark, and at 35% it becomes lethal.
- Cost - The current market price for deuterium is about US$300 per liter - much more expensive than gasoline. Method is available to extract deuterium from water, but there is no incentive to produce them in bulk.
- Delivery - Another problem is to deliver the isotopes to the specific sites. Something called "ifood" has been developed to do the job. It adds the isotopes to the essential amino acids not produced by the body, the reinforced amino acids will thus incorporate into the proteins. Another possibility is to feed deuterated water or isotope-enriched amino acids to farm animals.
- Ageing Causes - It is now understood that ageing has multiple underlying causes. Free radical damage alone cannot account for all the biological changes that happen with old age. It is doubtful that drinking deuterium laced water alone would stop ageing altogether.
An article in "New Scientist, May 16-22, 2015" reports that "Heavy Fat" metabolized in yeasts make them 150 times as resistant to ROS as those given regular fatty acid. The "Heavy Fat" chemical structure is identical to the regular one except that the hydrogen atoms are replaced by deuterium. A trial with the similar substance on patients with Friedreich's ataxia (this disease is caused by free radical damage to the nerves responsible for movement) will be launched in June, 2015.
 |
|
Figure 07a Telomere etc. |
years younger than people who did under 15 minutes. It has been known for some time that the telomerase enzyme, which is present naturally in some mammalian cells, can prevent the telomeres from unraveling. |
 |
|
Figure 07b Ageing Caused by Worn-out Cells [view large image] |
There are many methods proposed to deal with the problem including : |
- Senescent Cell Elimination - Destroying senescent cells by drug has been tested on mice. Two drugs for inducing cancer cells to commit suicide are identified as effective in killing senescent cells. It significantly delayed the onset of age-related conditions such as hunchback and cataracts. They are also stronger, looked younger. But it may not be a good idea to kill all the senescent cells because some of them do perform useful functions such as stopping tumour formation, wound healing, and embryo development.
- Stopping the Secretion of Harmful Chemicals - It is found that an existing drug called Rapamycin can do the job expanding the lifespan of mice by about 10%. However, this drug also weakens the immune system and been linked to increasing risk of diabetes. There are on-going researches to find the Rapamycin-like substance without such side effects.
- Limiting the Impact of Inflammation - There are many ways to alleviate the effect of inflammation including painkillers, food restriction, regular exercise, ... mostly involving the change of lifestyle (to healthier habits).
There are social implications on extending lifespan. Some researchers on ageing suggest that the effort is on staving off age-related diseases, relieving the suffering. Eventually, we will die from the depletion of stem cells as demonstrated by the blood sample of an 115 years old Dutch woman, who had only 2 left at the end while a healthy person would have about 1500 such cells. See original article on "Secret to Old Age Health" in New Scientist, 16 September 2015.
 |
These mice develop signs of premature ageing and die aged about a year instead of 2-3 years. The finding strongly support the idea that mutations in mitochondrial DNA can cause at least some features resembling ageing instead of just correlative (the manifestations of growing old). Figure 08a compares the mutating mice (mut) with the littermate wild-type (wt) at the age of ~ 40-45 weeks. It shows the mtDNA-mutator mice with reduced body size, kyphosis (curving of the spine), reduced hair density and different stages of alopecia (baldness) - delineated by dashed line (from bottom to top). New study on human have linked unhealthy mitochondria to Alzheimer's, Parkinson's, type 2 diabetes and other degenerative diseases. |
Figure 08a Mutating Mice [view large image] |
An unified theory of ageing is proposed in 2011 linking telomere damage in the cell nucleus to mitochondrial dysfunction.
 |
It is found that activation of the p53 protein in response to telemere damage can have different effects. In proliferative cells, p53 halts both cell growth and DNA replication, potentially causing apoptotic cell death (it also kills mutant cells). The 2011 new report indicates that p53 also represses the expression of PGC-1 genes in mitochondria, reducing the function and number of these organelles, and so leading to age-related dysfunction of mitochondrion-rich, quiescent tissues. This derangement also leads to the generation of toxic intermediates such as reactive oxygen species (ROS), which produce further |
Figure 08b Unified Theory of Ageing [view large image] |
damage in mitochondrial DNA setting up a vicious cycle of mitochondrial dysfunction (Figure 08b). Thus many of the causes for ageing are related through the action of p53. Paradoxically, this cancer eliminating protein also carries the penalty of ageing. |
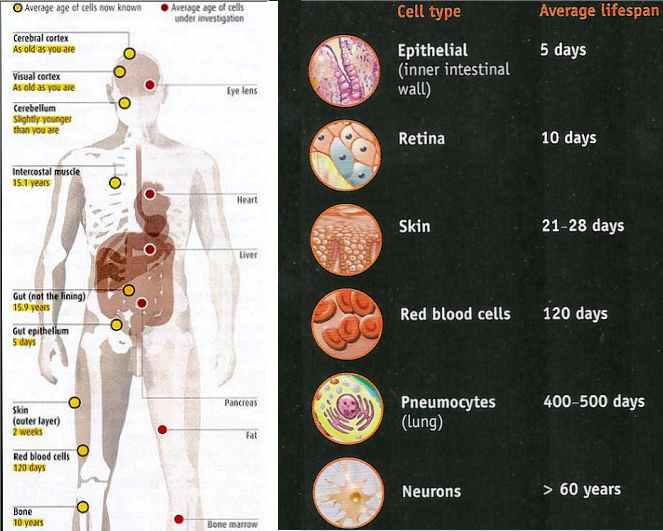 |
It is reported in 2006 that a new method enables the determination of aging in different parts of the human body. It is found that the body's front-line cells endure the roughest life, last the briefest time and are constantly replaced - these include the epithelial cells lining the gut (five days), the epidermal cells covering the skin's surface (2 weeks). The left diagram of Figure 08c shows the average age of other types of cell (yellow circles); those indicate by red circles are still under investigation. Now if the skin is so young, why don't we retain a smooth complexion even into old age? The answer lies with the mtDNA, which accumulates mutations at a faster rate than DNA in the nucleus. In skin, for instance, mitochondrial mutations are thought to be responsible for the gradual loss in the quality of collagen, the skin's scaffolding, which is why skin loses its shape and forms wrinkles. If we ever find ways to protect or repair mtDNA, this new discovery means that |
Figure 08c Aging of Organs |
we could significantly delay ageing. The diagram on the right of Figure 08c is a 2013 update on the aging of organs. |
 |
 |
|
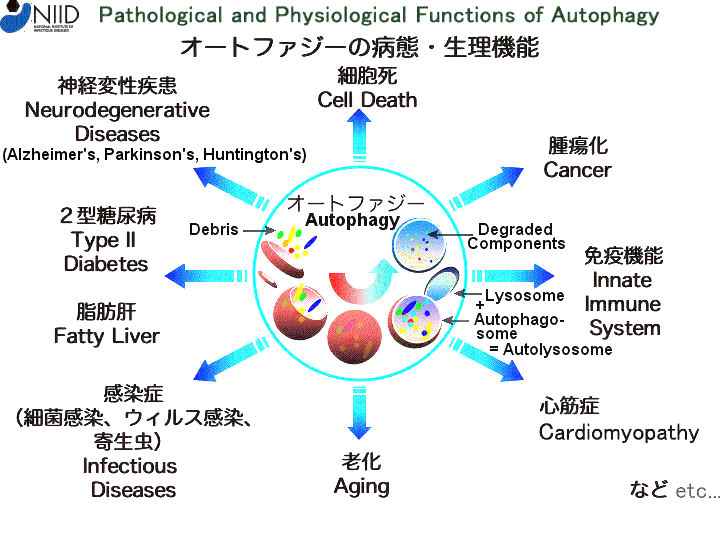
Figure 08d Autophagy, Process |
Figure 08e Autophagy, Malfunction |


