T = 16(p1/p2)2 e-2p2a/
for p2a/
 >> 1, where p1 = (2mE)1/2, and p2 = [2m(V-E)]1/2.
>> 1, where p1 = (2mE)1/2, and p2 = [2m(V-E)]1/2.
 |
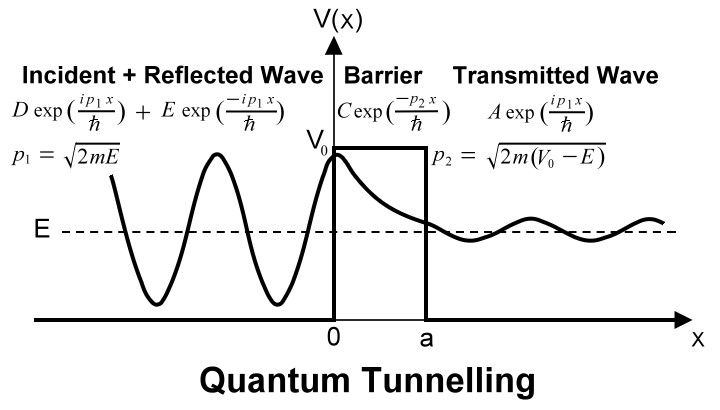 |
This formula provides a crude approximation in estimating the probability of alpha decay, which depends inversely on the height "V" and width "a" of the barrier. Alpha decay is a process of asymmetric fission because it usually involves a larger nuclear fragment and the much smaller alpha particle. |
Figure 14-11a Nuclear Potential [view large image] |
Figure 14-11b Quantum Tunneling [view large image] |
92U235 + n
 92U236
92U236  56Ba144 + 36Kr89 + 3n + 166 Mev
56Ba144 + 36Kr89 + 3n + 166 Mev
 |
 |
For uranium-238, the excitation energy is about 1 Mev less, so fission is not possible with slow neutrons; it can take place only for neutrons with 1 Mev energy or more. An isotope like uranium-235 that can be split by both slow and fast neutrons, is called "fissile", while uranium-238 which can be split only by fast neutrons, is called "fissionable". |
Figure 14-11c Neutron Induced Fission and Chain reaction [view large image] |
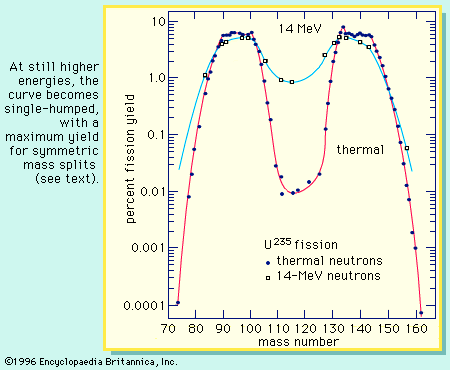
Figure 14-11d Symmetric Fission [view large image] |
 |
|
Figure 14-11e Fission Cross Section [view large image] |
fission cross section of 640x10-24cm2 for slow neutron (at 0.025 ev) is due to quantum effect. While the threshold of 1 Mev for U238 has been explained already above. |
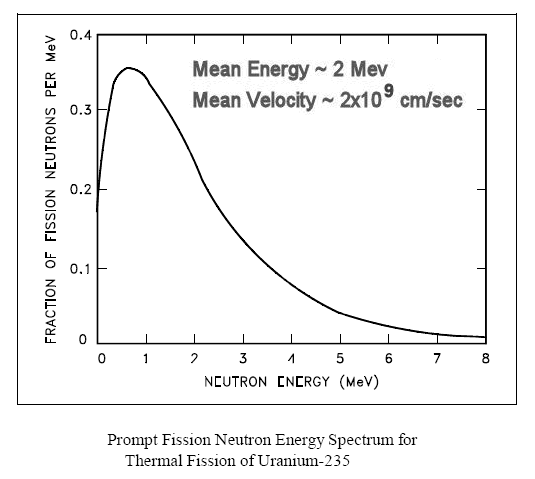 |
Therefore, chain reaction can be maintained only when the diameter of the sphere is at least equal to that length. From the known density of 18.9 gm/cm3 for U235, the critical mass is 44 kg. The arithmetic to produce all these numbers is very simple via the formula: M =  [(4 [(4 /3)R3] /3)R3]where  is the density, R the radius, and M the mass of the isotope. The time scale can be estimated from: (# of mean-free-path)/(average velocity of fast neutrons ~ 2x109cm/sec). See Figure 14-11f for the neutron spectrum from the fission process. Prompt neutron is the one released together with the fission. is the density, R the radius, and M the mass of the isotope. The time scale can be estimated from: (# of mean-free-path)/(average velocity of fast neutrons ~ 2x109cm/sec). See Figure 14-11f for the neutron spectrum from the fission process. Prompt neutron is the one released together with the fission.
|
Figure 14-11f Neutron Spectrum [view large image] |
| Fissionable Isotope | Crtiical Mass (kg) | Half Life (years) | Neutron Generation (# / sec-kg) |
Power Generation (Watts / kg) |
|---|---|---|---|---|
| Protactinium-231 | 162 | 3.28x104 | nil | 1.3 |
| Thorium-232 | Infinite | 1.41x1010 | nil | nil |
| Uranium-233 | ~ 8 | 1.59x105 | 1.23 | 0.281 |
| Uranium-235 | 47.9 | 7.0x108 | 0.364 | 6x10-5 |
| Uranium-238 | Infinite | 4.5x109 | 0.11 | 8x10-6 |
| Neptunium-237 | 59 | 2.14x106 | 0.139 | 0.021 |
| Plutonium-238 | 10 | 88 | 2.67x106 | 560 |
| Plutonium-239 | 10.2 | 2.41x104 | 21.8 | 2.0 |
| Plutonium-240 | 36.8 | 6.54x103 | 1.03x106 | 7.0 |
| Plutonium-241 | 12.9 | 14.7 | 49.3 | 6.4 |
| Plutonium-242 | 89 | 3.76x105 | 1.73x106 | 0.12 |
| Americium-241 | 57 | 433 | 1540 | 115 |
| Americium-242 | 9 - 18 | - | - | - |
| Americium-243 | 155 | 7.38x103 | 900 | 6.4 |
| Curium-244 | 28 | 18.1 | 1.1x1010 | 2.8x103 |
| Curium-245 | 13 | 8.5x103 | 1.47x105 | 5.7 |
| Curium-246 | 84 | 4.7x103 | 9x109 | 10 |
| Curium-247 | 7 | 1.55x107 | - | - |
| Berkelium-247 | 10 | 1.4x103 | nil | 36 |
| Californium-251 | 9 | 898 | nil | 56 |