


The silicon atom looks like carbon in the periodic table, its larger size, weaker bonds, and chemical preferences make it unsuitable for building the
| Home Page | Overview | Site Map | Index | Appendix | Illustration | Preface | Contact | Update | FAQ |
 |
 |
Its 4 valence electrons can either capture or donate 4 more as shown below ; The silicon atom looks like carbon in the periodic table, its larger size, weaker bonds, and chemical preferences make it unsuitable for building the |
Figure 01 Periodic Table, |
Figure 02 Periodic Table, |
complex, stable, and versatile molecules needed for life. Carbon, with its Goldilocks bonding properties, remains unmatched in that role. |

 |
 |
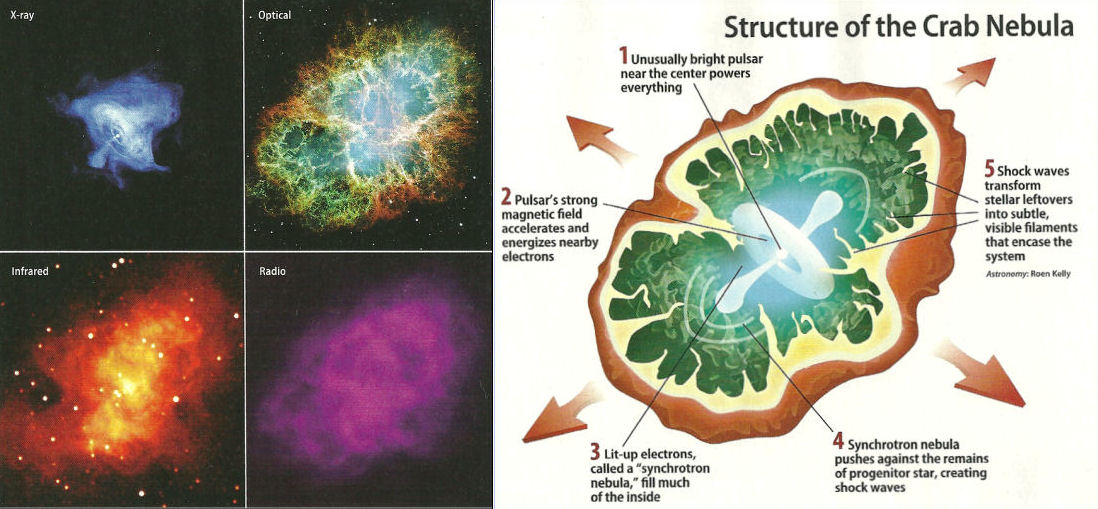 |
|
Figure 03 Periodic Table, Origin |
Figure 04 Production in Supergiants |
Figure 05 Dispersal by Supernova |


 ________________________________________
________________________________________ ________________________________________
________________________________________
 ________________________________________
________________________________________