 |
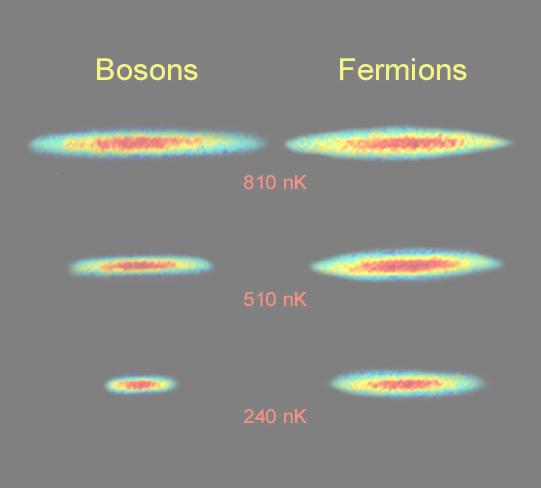
The Pauli Exclusion Principle states that identical fermions -- one type of fundamental matter with 1/2 integer spin quantum number -- cannot be in the same place at the same time and with the same orientation (i.e., cannot have the same quantum state). It is this Exclusion Principle that requires the electrons in an atom to occupy different energy levels instead of all congregating in the lowest energy level. Chemistry would be very different without this rule. The exclusion principle is also responsible for the degenerate pressure, which prevents the White Dwarf from complete collapse. The other type of matter, bosons (particles with integer spin quantum number), do not have this property. The boson gas can form Bose-Einstein condensate near absolution zero temperature. They are all in the same quantum state, and behave like a single entity. Figure 12-04a shows the bosons bunch together, while the fermions keep their distance at temperature a few hundreds billionths of a degree above absolution zero (nanoKelvin=nK). Superfluidity refers to frictionless flow of spin 0 bosons, e.g., helium-4 at low temperature.
|

