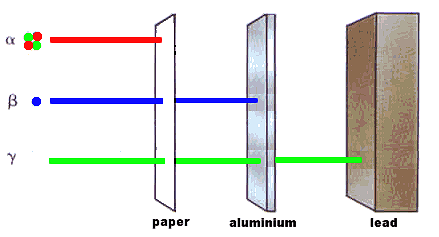
Unstable nuclei, called radioactive isotopes, will undergo nuclear decay to make it more stable. There are only certain types of nuclear decay which means that most isotopes can't jump directly from being unstable to being stable. It often takes several decays to eventually become a stable nucleus. When unstable nuclei decay, the reactions generally involve the emission of a particle and or energy. Half-lives are characteristic properties of the various unstable atomic nuclei and the particular way in which they decay. Alpha and beta decay are generally slower processes than gamma decay. Half-lives for beta decay range upward from 10-2 sec and, for alpha decay, upward from about 10-6 sec. Bismuth-209 has the longest half-life of 2x1019 years. Half-lives for gamma decay may be too short to measure (~ 10-14 second), though a wide range of half-lives for gamma emission has been reported.
| Type |
Emission |
Penetrating Power |
Example |
| Alpha Decay |
Helium nuclei |
1, stopped by skin, very damaging due to ionization |
92U238  90Th234 + 2He4 90Th234 + 2He4
Applicable to nuclei with Z>82, see Figure 14-02 |
| Beta Decay |
Electron, high speed |
100, penetrates human tissue to ~ 1 cm |
53I131  54Xe131 + -1e0 54Xe131 + -1e0
Applicable to nuclei with high neutron-proton ratio |
| Gamma Decay |
Photons, high energy |
10000, highly penetrating but not very ionizing |
92U238  90Th234 + 2He4 + 2 photon 90Th234 + 2He4 + 2 photon
Energy lost from settling within the nucleus after transmutation |
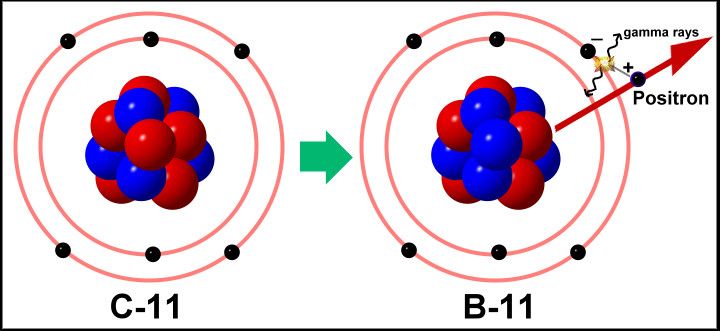
| Positron Emission |
Positron |
100 |
6C11  5B11 + 1e0 5B11 + 1e0
Applicable to nuclei with a low neutron-proton ratio |
| Electron Capture |
Neutrino (from inner shell -1e0) |
~ Infinite for Neutrino |
37Rb81 + -1e0  36Kr81 + neutrino ( 36Kr81 + neutrino ( e) e)
Applicable to nuclei with a low neutron-proton ratio |


 p + e + electron-anti-neutrino with a lifetime about 15 minutes in a process known as beta decay. Neutrons in a nucleus are subject to the protection of the nuclear and the electromagnetic forces from the other nucleons, and they will remain stable provided there are not too many of them. If there are too many, such protection would not be sufficient for all of them to remain stable, and the nucleus would undergo beta decay. Figure 14-07 shows the beta decay process, in which the down quark turns into an up quark (thus changes the neutron to proton) by emitting a W- meson, which decays into an electron and an electron-anti-neutrino.
p + e + electron-anti-neutrino with a lifetime about 15 minutes in a process known as beta decay. Neutrons in a nucleus are subject to the protection of the nuclear and the electromagnetic forces from the other nucleons, and they will remain stable provided there are not too many of them. If there are too many, such protection would not be sufficient for all of them to remain stable, and the nucleus would undergo beta decay. Figure 14-07 shows the beta decay process, in which the down quark turns into an up quark (thus changes the neutron to proton) by emitting a W- meson, which decays into an electron and an electron-anti-neutrino. 

 e)
e)